Clinical data management (CDM) is an important phase in clinical research, which leads to having high-quality and reliable data. It has the main four components i.e. data integrity, data quality, data security and data privacy.
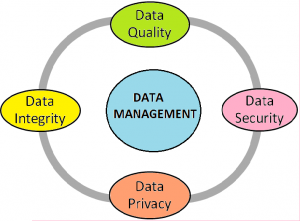
CDM is a combination of data collection, cleaning, and management in compliance with regulatory standards. The main aim of CDM processes is to provide high-quality standard data by reducing errors and missing data. A high-quality data should meet the protocol-specified parameters, comply with the protocol requirements and absolutely accurate & suitable for statistical analysis as per Statistical Analysis Plan (SAP).
CMD process begins with protocol development to the database locking. The process is self-explanatory in Figure1.
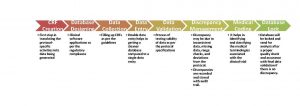
Adoption of a process shown above and standard instruments/tools with responsible CDM team will provide you high-quality and validated data for clinical research which results in high-quality research outcome.
Last modified: 21/11/2019






